I recently joined a Q Visit to Health Improvement Scotland (HIS) to learn about their work in developing a national Quality Management system and how this could help deliver high-quality care.
One delegate mentioned the difficulty of knowing where to focus improvement work. The conversation took me back to a recent visit to the Institute for Healthcare Improvement’s (IHI) Immersion programme which I attended as part of the Health Foundation GenerationQ Fellowship. A conversation during a workshop there came to mind.
This is exciting work but feels quite daunting. We are aware it will take years to get it right.
“How do I even know where to begin with improvement work?” One facilitator at the IHI had posed this question to an author of The Improvement Guide, an influential text outlining the Model for Improvement (MfI), a popular approach to Quality Improvement (QI). The facilitator had started to question how far the MfI could take you when trying to deliver whole-system improvement.
The author replied: “Well, I’d assume you’d have a good Quality Planning process and a good Quality Control system so you know how you’re doing”.
During the IHI programme and the visit to HIS we heard how QI works best as part of an improvement system consisting of three main elements:
- Quality Planning: figuring out what you want the system to deliver and how to make that happen
- Quality Control: a process of monitoring and adjusting the system to deliver the desired outcomes
- Quality Improvement: using a systematic method to improve performance and quality
Together these form what is known as a Quality Management System (QMS). HIS have shared their learning from a 90-day innovation cycle as well as other useful documents on their website.
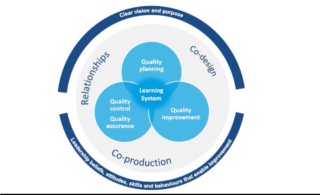
A useful overview of East London Foundation Trust approach and tips around building a QMS is shown in Figure 2.
During the day at HIS, sessions focused on the three aspects of QMS as well as looking at the conditions that support the system. Many interesting issues emerged; I will highlight three.
Firstly, the role of quality assurance in health care which consists both of internal and external monitoring and inspection. Is it part of Quality Control? Ideally, assurance and control measures are closely aligned to prevent stakeholders from looking at different metrics to understand how a system is functioning. I have found that often the quality metrics that frontline clinicians, commissioners and inspectors are interested in can be quite different. Assurance processes tend to be more sporadic and quality control sit closer to the point at care delivery.
Secondly, HIS have outlined a set of behaviours that underpin the different phases such as focusing on collaboration and co-design and making these essential aspects of the design process in each phase. It showed me how wh

at can be conceived as a mechanistic design process which came from industry can be used to help promote certain values and cultural norms. QMS can be as much about promoting collaboration in design and health care, an essential part of building Integrated Care Systems.
Finally, it laid bare the challenge of informatics and getting the right data, essential in developing a learning organisation. I was struck by how challenging it was to co-ordinate data on surgical morbidity and mortality from different organisations. They had worked hard at reducing the number of key metrics (from over a hundred to just a handful).
In my own early experimentation with building a small-scale QMS, agreeing the set of measures that can help guide teams, ensuring this aligns with other stakeholders, ensuring the data quality is good and then making it accessible and feel like it is owned by teams at the point of care is a real challenge.
Like many trusts in the UK, my organisation has developed a Quality Improvement (QI) team over the past few years, many of our staff have trained in QI methodology, lots of projects are happening and there has been real excitement from leadership about the potential of QI in providing better care in challenging circumstances. Quality improvement, Don Berwick points out, in Curing Healthcare is where most places start.
While I have seen great work and clear improvement at a team (or ‘micro-system’) it is harder to feel confident about the impact across the whole system and on whole-system outcomes. This step, from micro-system to whole-system improvement feels huge.
HIS have outlined a set of behaviours that underpin the different phases such as focusing on collaboration and co-design and making these essential aspects of the design process in each phase.
The Health Foundation publication The Improvement Journey provides a great overview of this challenge and thoughts about how to make the leap. It highlights the importance of aligning activity across the system and that teams focusing on their pet improvement project can detract from the system as a whole improving. I think this is what Gary Kaplan (Virginia Mason CEO) was talking about at a recent International Forum when he said the QI projects could be damaging to an organisation. That is, energy and enthusiasm being spent locally without attention to whole-system problems, and spread and scale-up of improvement work.
In essence, this feels like what a QMS in health care is about – purposeful design of each part of the system, understanding how each micro-system can help achieve common goals, creating shared purpose where improvement activity can be co-ordinated and amplified.
In my own organisation our Trust Board recently approved our proposal to start development of a QMS. We are developing some prototypes with individual teams, and are working together with our informatics team, performance committees and Quality Governance so that we can build the data system and be clear about all the competing demands of the teams involved. We have also begun mapping out how we can create a Board-to-Floor golden thread that helps each part of the system understand how they contribute to a shared outcome.
This is requiring our QI team to work more closely with many other parts of the system, to understand the demands and expectations of each part of the system. At the same time we are trying to allow the teams themselves a strong voice in building their own QMS with metrics that matter to them and their service users.
We have also been trying to develop a stronger QI presence at senior strategic and operational levels to support Quality Planning work, drawing in frontline teams into this work and agreeing together on what the system is hoping to achieve, and how we can deliver this. Historically there has been a disconnect between planning at senior level, where Key Performance Indicators are negotiated with commissioners, excluding the teams delivering the services who should be overseeing quality control but who are disinvested by not being involved in the process.
This is exciting work but feels quite daunting. We are aware it will take years to get it right. But if we do I think we will be closer to realising the potential that QI has beyond the team-level to improving the whole system.
Comments
Isabel Ho 15 Jan 2020
Hi Fredrik, very interesting read for me. In our discipline of radiation oncology qms is mandatory by service specification. We ve been using qms for few decades and have very well established frameworks compared to other disciplines.
One thing concerns me is the use of qms. For us it refers to ISO9001 qms standards which is recognized internationally across industries including healthcare but the qms you refer to have a different focus, howeve, we can argue the qms you're referring to is the next evolution of qms standards?! It is worth highlighting this to your readers as we are not necessaily talking about the same thing although the underlying common purpose is possibly identical/similar.
Best wishes
Isabel
Fredrik Johansson 24 Jan 2020
Hi Isabel,
Thanks for the comment - you raise some really interesting issues.
Firstly, I agree that there are probably some differences in what people mean when they talk about QMS and you are right in pointing out the importance of defining this. It seems to me that organisations and systems are interpreting this in their own way which might lead to confusion and misunderstanding - such a wide view of what co-production means nowadays and much is far from the original description.
Secondly I was not aware that there were some widely recognised industry standards for QMS so will be looking at this for learning. I was also interested to hear how long radiation oncology has been using QMS and would be really interested if you might share more about how this has worked in your field of health care and what can be applied to other settings from this.
Thanks again.
Hans 4 Oct 2020
Not spamming but just wanted to say this is really
useful!
Ahamed Basheer 22 Dec 2020
Very highly informative
Thomas John Rose 21 Mar 2021
In the UK we have been managing quality for over 100 years. The UK's Chartered Quality Institute began on 1st April 1919. I can't understand why the NHS insist on following America when it comes to managing quality. The UK is much better at it! Isabel's QMS is a UK QMS.
Isabel Ho 21 Mar 2021
Hi Tom, interesting I didn't think this way before. In fact CQI is one of the key element in any ISO QMS and UK (BSI) has been leading on this for many years. Instead of reinventing the wheel, I suppose we can see this as a targeted drive or focus on CQI by telling the story with different narrative for the health & care audiences?